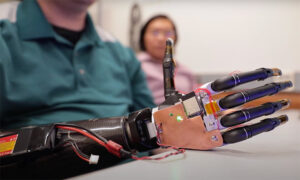
The US Food and Drug Administration granted breakthrough device designation to two products developed by med-tech startup Fasikl. The company’s MindForce nerve-computer-interface system gives people with amputations control of a robotic hand or computer device, and Epione alleviates post-amputation pain.
Use of the products requires minimally invasive surgery to insert an implantable neural recorder into a person’s residual limb than can then be connected through Bluetooth to a computer or phone, which makes them Class III devices requiring premarket approval.
One of the participants in the product testing, Cameron Slavens, said he was able to use Fasikl’s prosthetic arm with very few adjustments, and it helped relieve his residual limb and phantom limb pain. Slavens told Twin Cities Business that his pain is still minimal years later.
CEO Zhi Yang, PhD, anticipates securing full FDA approval within ten years. Fasikl has 30 employees and has raised $17 million.
To learn more about the products, visit “AI Gives More Accurate, Less Invasive Prosthetic Control.”




